Reimbursement and Coverage
For HCPs / Reimbursement and Coverage
Patients may avail of CHEMOSAT in one of our 22 authorised centres throughout Europe.
To view the centres please click here.
Access to treatment will depend on whether CHEMOSAT is covered through a reimbursement code by a national payer or by private health insurance. Patients can also opt to self-pay if other options are unsuccessful.
Private health insurance
Private patients should apply to their private health insurer provider to enquire about accessing funding towards CHEMOSAT. To access CHEMOSAT privately, the patient's medical oncologist will need to send a referral letter to the treating centre, documenting the medical condition, as well as providing a history of recent scans, histology and previous treatments provided.
Self-pay
If the patient's national payer or private health insurance does not support CHEMOSAT, patients have the option of self-pay. Patients can ask their medical oncologist to send a referral letter to an authorised CHEMOSAT centre for review by the treating team. In this instance the cost of the treatment must be fully supported by the patient.
For country specific details, please see below
In Germany, CHEMOSAT is funded through a combination of a diagnostic related group (DRG) code and an additional fee under the Zusatzentgelt (ZE). This funding permits hospitals in Germany to obtain reimbursement for CHEMOSAT procedures (in effect since 2016).


National Institute for Health and Care Excellence (NICE) has updated its guidance for the use of CHEMOSAT in the treatment of patients with metastaic uveal melanoma. Under the revised NICE guidance (changed from a research-only designation), CHEMOSAT has been categorised under a Special Arrangements designation. Under this designation, the number of private health insurance companies funding CHEMOSAT should increase, and some regional funding may be more accessible.
Oncologist
A medical oncologist will usually be responsible for ensuring that CHEMOSAT is the right treatment approach for their patient. They are experienced in the monitoring of toxicities of chemotherapy and are responsible for the complete medical management of the patient, including, but not limited to, pre- and postoperative care. They may also be responsible for monitoring the patient during the immediate post-procedure period.
Intensivist/critical care specialist
A qualified intensivist, or appropriately qualified critical care specialist, is responsible for providing medical management of the patient in the immediate post-procedure period, during which the patient is in the intensive care unit or step down unit.
lnterventional radiologist
A qualified interventional radiologist with the knowledge, skills, experience, and hospital privileges required to perform advanced vascular interventional procedures.
Nurse
A qualified chemotherapy healthcare professional certified by the site to deliver chemotherapy, such as Interventional Radiology Technician or Registered Nurse.
Perfusionist
A qualified perfusionist is required to establish, monitor, and control the extracorporeal pump and veno-venous bypass circuit.
Anaesthesiologist
A qualified anaesthetist (anaesthesiologist) and/or nurse anaesthetist responsible for the management of sedation, analgesia, respiratory and cardiovascular support.
Pharmacist
A qualified pharmacist, on call during the procedure, to reconstitute the chemotherapeutic agent (melphalan hydrochloride), using national and local safety guidelines. The pharmacist should be aware of the rapid preparation time required for the preparation and administration of melphalan for use with CHEMOSAT.
Assess Eligibility 7
*Patients with 50% or greater tumour burden of the liver on imaging should have a laparoscopy and normal liver biopsied for both visual inspection of liver parenchyma and histological examination of the liver to rule out hepatocellular dysfunction at a subclinical level
Clinical Assessment
Assessment by Expert CHEMOSAT Team
Post Treatment Assessment
Week 1 - blood tests
Week 2 - blood tests
Week 3 - blood tests
Week 6-8 - post treatment MRI to assess response
Thank you for your interest in CHEMOSAT.
The following pages are exclusively reserved for health care professionals licensed in the European Union, the United Kingdom, Switzerland, Norway, Iceland, Turkey & Liechtenstein where the product has been CE marked.
If you are located outside of these regions, please click here (www.delcath.com)
Information provided on this site is not intended to be professional medical advice. Product Instructions for Use (IFU) should be consulted before use of the product.
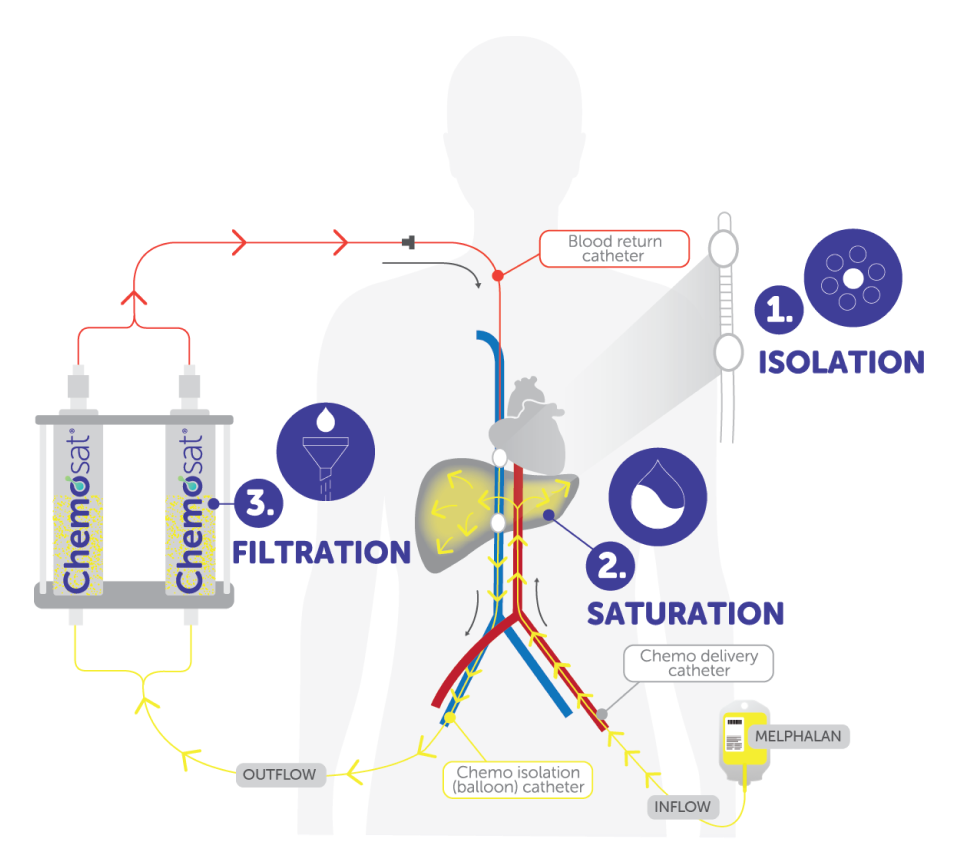
I UNDERSTAND AND AGREE TO THE STATEMENT ABOVECHEMOSAT: A 3 Step Process
Czech Republic
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 26-JUL-2023 | 120056 Rev E | Czech • Download |
| 26-JUL-2023 | 120056 Rev E | English • Download |
| 21-APR-2022 | 120056 Rev D | Czech • Download |
| 21-APR-2022 | 120056 Rev D | English • Download |
| 08-MAR-2021 | 120056 Rev C | Czech • Download |
| 08-MAR-2021 | 120056 Rev C | English • Download |
France
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 22-JAN-2024 | 120057 Rev F | French • Download |
| 22-JAN-2024 | 120057 Rev F | English • Download |
| 26-JUL-2023 | 120057 Rev E | French • Download |
| 26-JUL-2023 | 120057 Rev E | English • Download |
| 21-APR-2022 | 120057 Rev D | French • Download |
| 21-APR-2022 | 120057 Rev D | English • Download |
| 08-MAR-2021 | 120057 Rev C | French • Download |
| 08-MAR-2021 | 120057 Rev C | English • Download |
Germany
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 26-JUL-2023 | 120055 Rev F | German • Download |
| 26-JUL-2023 | 120054 Rev F | English • Download |
| 21-APR-2022 | 120055 Rev E | German • Download |
| 04-APR-2022 | 120054 Rev E | English • Download |
| 08-MAR-2021 | 120055 Rev D | German • Download |
| 08-MAR-2021 | 120054 Rev D | English • Download |
Italy
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 26-JUL-2023 | 120056 Rev E | Italian • Download |
| 26-JUL-2023 | 120056 Rev E | English • Download |
| 21-APR-2022 | 120056 Rev D | Italian • Download |
| 21-APR-2022 | 120056 Rev D | English • Download |
| 08-MAR-2021 | 120056 Rev C | Italian • Download |
| 08-MAR-2021 | 120056 Rev C | English • Download |
The Netherlands
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 26-JUL-2023 | 120055 Rev F | Dutch • Download |
| 26-JUL-2023 | 120054 Rev F | English • Download |
| 21-APR-2022 | 120055 Rev E | Dutch • Download |
| 04-APR-2022 | 120054 Rev E | English • Download |
| 08-MAR-2021 | 120055 Rev D | Dutch • Download |
| 08-MAR-2021 | 120054 Rev D | English • Download |
Portugal
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 26-JUL-2023 | 120056 Rev E | Portuguese • Download |
| 26-JUL-2023 | 120056 Rev E | English • Download |
| 21-APR-2022 | 120056 Rev D | Portuguese • Download |
| 21-APR-2022 | 120056 Rev D | English • Download |
| 08-MAR-2021 | 120056 Rev C | Portuguese • Download |
| 08-MAR-2021 | 120056 Rev C | English • Download |
Spain
| Publication Date | Document and Revision number | Language |
|---|---|---|
| 22-JAN-2024 | 120057 Rev F | Spanish • Download |
| 22-JAN-2024 | 120057 Rev F | English • Download |
| 26-JUL-2023 | 120057 Rev E | Spanish • Download |
| 26-JUL-2023 | 120057 Rev E | English • Download |
| 21-APR-2022 | 120057 Rev D | Spanish • Download |
| 21-APR-2022 | 120057 Rev D | English • Download |
| 08-MAR-2021 | 120057 Rev C | Spanish • Download |
| 08-MAR-2021 | 120057 Rev C | English • Download |
References
References
References
References
Cookies Policy
Using our website
Our website (“chemosat.com”) uses cookies to improve your experience and to provide tailored information.
What are cookies?
Cookies are text files stored on your computer so that when you visit a website it can remember who you are. Using cookies allows websites to remember your preferences and behaviour which enables websites to improve your experience.
Cookies can be set by the owner of the website or in some cases by third-party services the website owner allows to present or store other information or provide other functionality such as data analytics.
How are they used on this site?
Cookies are used to improve the functionality of our website.
This website uses session cookies. Session cookies are used to deliver the basic functions of a website i.e. to allow pages to remember technical changes or selections you may make between pages. Session cookies are temporary cookies and are generally erased when you close your browser.
We may also use cookies set by third parties for other functionalities such as data analytics and improved targeted advertising.
Managing & Removing Cookies
Should you change your mind in the future you always have the option of removing cookies from your browser. Please the below links depending on your browser: